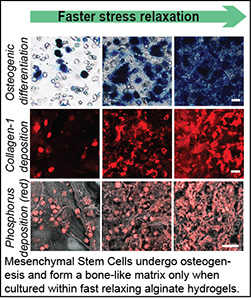
Many soft tissues are viscoelastic, exhibiting stress relaxation in response to a deformation. We have found that extracellular matrix viscoelasticity regulates various cellular behaviors, such as spreading, differentiation, matrix formation, matrix remodeling, migration, and division. We seek to how elucidate the mechanisms by which matrix viscoelasticity influences cell biology, and harness these insights towards the development of new therapies and applications in regenerative medicine. A key finding is that cells utilize stretch-activated ion channels to sense matrix viscoelasticity.
Relevant publications: 1. Lee, Stowers, and Chaudhuri, Volume expansion and TRPV4 activation regulate stem cell fate in three-dimensional microenvironments , Nature Communications (2019). 2. Lee, et al, Mechanical confinement regulates cartilage matrix formation by chondrocytes , Nature Materials (2017). 3. Chaudhuri, et al., Hydrogels with tunable stress relaxation regulate stem cell fate and activity, Nature Materials (2016). 4. Chaudhuri, et al., Substrate stress relaxation regulates cell spreading, Nature Communications (2015).
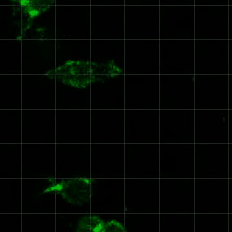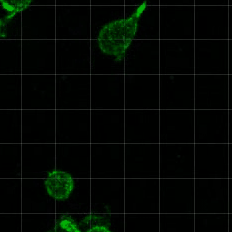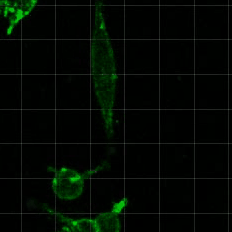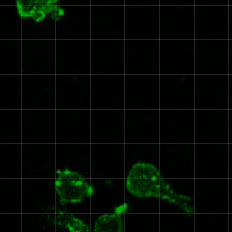
Cell migration is critical for development, immune cell trafficking, wound healing, and metastasis. In many contexts, cells migrate through three-dimensionally confining microenvironments, where extracellular matrix pore size is smaller than that of the cells. For example, during cancer progression, carcinoma cells on the order of 10 microns in size must invade through a nanoporous basement membrane as a first step towards metastasis. We are investigating how cells push, pull, and degrade matrix in order to migrate through 3D tissue microenvironments. A particular area of interest is in understanding the role of matrix mechanical plasticity in mediating invasion and migration of cancer cells.
Relevant publications: 1. Wisdom, et al, Matrix mechanical plasticity regulates cancer cell migration through confining microenvironments , Nature Communications (2018).2. Wisdom, et al, Covalent cross-linking of basement membrane-like matrices physically restricts invasive protrusions in breast cancer cells, Matrix Biology (2019).
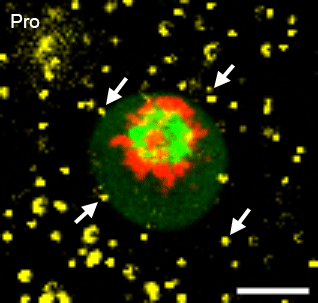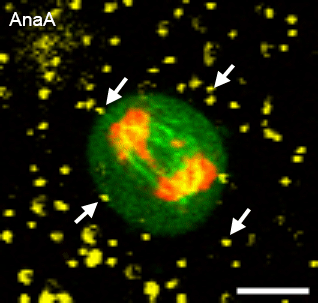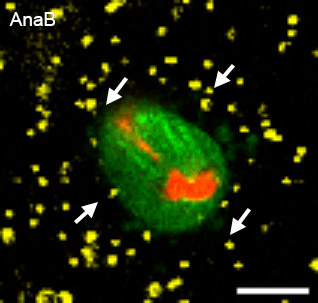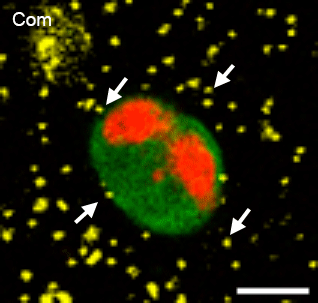
In many physiological contexts, cells divide in mechanically confining viscoelastic microenvironments, where they are surrounded by neighboring cells and ECM. Most of what is known about cell division is based on studies of cells dividing on 2D substrates, where they can simply release from the substrate and divide unrestricted. A mechanically confining microenvironment would be expected to restrict various morphological processes associated with cell cycle progression and cell division. We aim to uncover the how confinement impacts cell cycle progression, and how cells generate forces in order to divide in confining microenvironments.
Relevant publications: 1. Nam and Chaudhuri, Mitotic cells generate protrusive extracellular forces to divide in three-dimensional microenvironments , Nature Physics (2018).2. Nam, et al. Cell cycle progression in confining microenvironments is regulated by a growth-responsive TRPV4-PI3K/Akt-p27Kip1 signaling axis, in press.
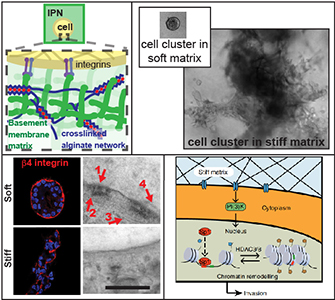
Increased mammographic density, associated with an increase in tissue stiffness, is one of the strongest and most consistent risk factors for breast cancer progression. Various studies have established that increased stiffness promotes cancer progression, but the mechanisms mediating the effect of stiffness remain unclear. We seek to understand the mechanisms underlying the impact of increased stiffness on breast cancer progression in terms of cell-matrix interactions, and transcriptional and epigenetic regulation. Our vision is that by better understanding how mechanics influences breast cancer progression, we can develop more effective diagnostics and therapies.
Relevant publications: 1. Stowers, et al. Matrix stiffness induces a tumorigenic phenotype in mammary epithelium through changes in chromatin accessibility, Nature Biomedical Engineering (2019). 2. Lee, et al. YAP-independent mechanotransduction drives breast cancer progression, Nature Communications (2019). 3. Chaudhuri, et al. Extracellular matrix stiffness and composition jointly regulate the induction of malignant phenotypes in mammary epithelium, Nature Materials (2014).
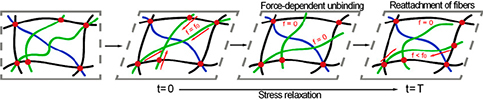
Many biological materials, from the actin cytoskeleton inside cells, which governs their shape and rigidity, to the type-1 collagen rich extracellular matrix, consist of semiflexible biopolymer networks with highly organized architectures and crosslinking interactions. These networks often have unique behaviors of nonlinear elasticity, viscoelasticity, plasticity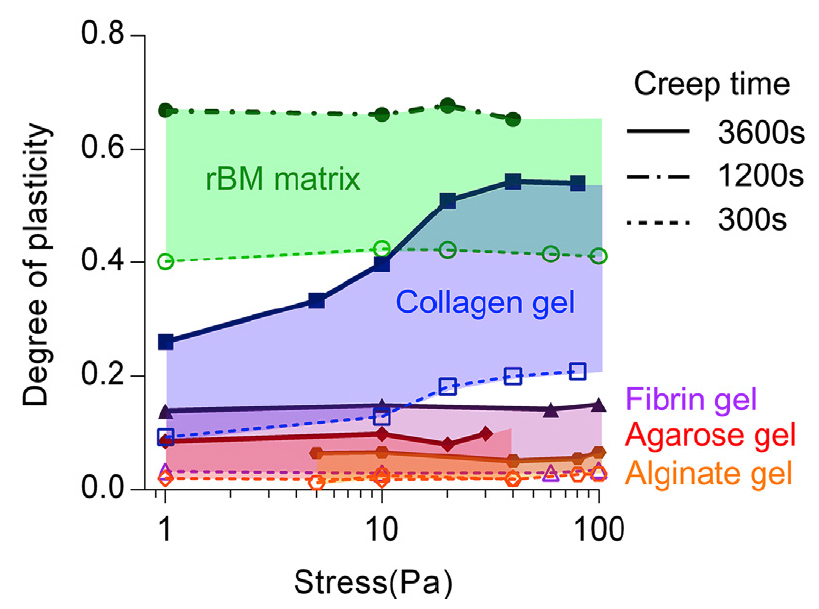 , and toughness but the molecular mechanisms underlying these behaviors are not fully understood. Our goal is to characterize these mechanical behaviors and elucidate theunderlying molecular mechanisms.
, and toughness but the molecular mechanisms underlying these behaviors are not fully understood. Our goal is to characterize these mechanical behaviors and elucidate theunderlying molecular mechanisms.
Relevant publications: 1. Nam, et al., Strain-enhanced stress relaxation impacts nonlinear elasticity in collagen gels, PNAS (2016). 2. Nam, et al., Viscoplasticity Enables Mechanical Remodeling of Matrix by Cells, Biophysical Journal (2016).
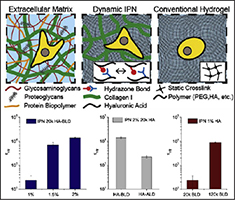
Hydrogels are often used as artificial extracellular matrices for cell culture studies and various applications in drug delivery and regenerative medicine. We are developing hydrogels with independently tunable mechanical properties for use in these applications.
Relevant publications: 1. Lou, et al., Stress Relaxing Hyaluronic Acid-Collagen Hydrogels Promote Cell Spreading, Fiber Remodeling, and Focal Adhesion Formation in 3D Cell Culture, Biomaterials (2018). 2. Nam, et al., Varying PEG density to control stress relaxation in alginate-PEG hydrogels for 3D cell culture studies, Biomaterials (2019).

Forces and mechanics at the micro and nanoscale play a key role in regulating cell-matrix interactions, but current approaches to measure these have limitations. Thus, there is a critical need for new tools to probe forces and mechanics at the micro and nanoscales. We are developing tools that provide new capabilities in this space.

